Autoignition mechanisms
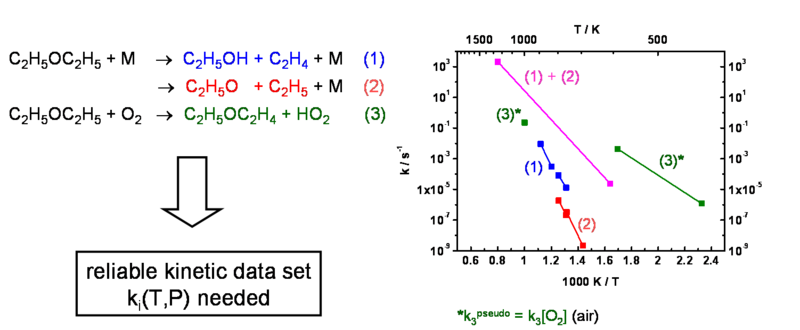
Diethyl ether has a low minimum ignition temperature and is often used to investigate safety-relevant ignition processes at hot surfaces. The ignition mechanism and the kinetic parameters even of the initial steps, however, are not or not sufficiently well known. Our project aims at determining missing kinetic and thermochemical data by different experimental and theoretical methods such as shock tube and laser photolysis experiments and quantum chemical and statistical rate theory calculations, respectively. At the beginning the emphasis will be put on pyrolytic reactions of diethyl ether itself, its reactions with molecular oxygen, and its reactions with intermediates like H atoms and OH radicals.
The final goal is the development of a reaction mechanism for diethyl ether ignition/combustion, which is valid over a wide range of temperature, pressure, and concentration, and which is supported by reliable kinetic data. The project will in particular collaborate with project 1, where the kinetic data are to be incorporated in a general model to characterize and predict the ignition behavior under a variety of different conditions.
Scientists
| Project leader | |
| PhD student | |
| PhD student |
Articles: | |
|---|---|
| 3. | Proc. Combust. Inst. (2014) |
| 2. |
J. Phys. Chem. A
117, 8343-8351
(2013)
DOI: 10.1021/jp405724a
|
Conferences: | |
| 1. |
Proceedings of the European Combustion Meeting 2013
, page P3-13.
June
2013
978-91-637-2151-9
|